A new study reports that tungsten carbide-based catalysts can hydrocrack polyolefin plastics efficiently even when the feed contains polyvinyl chloride, or PVC, a contaminant that often disables conventional recycling catalysts. The work points to a potentially important advance for chemical recycling, where real plastic waste streams are rarely pure and often contain additives, mixed polymers and traces of chlorine-bearing materials.
The research, published in the Journal of the American Chemical Society, was carried out by scientists from the University of Akron and collaborating institutions including the University of Rochester, Indiana University, the University of North Texas and Pacific Northwest National Laboratory. The team describes tungsten carbide, written as WxC, as an intrinsically bifunctional catalyst that combines metal-like and acid-like behavior in a single material platform.
That matters because hydrocracking of polyolefins depends on two different catalytic jobs happening together. One set of sites must carry out hydrogenation and dehydrogenation steps. Another set, known as Brønsted acid sites, must promote isomerization and carbon-carbon bond cleavage. In most established systems, those functions are supplied by separate components, often a noble metal combined with a zeolite acid support. Such catalysts can work well on purified feeds, but they are less suited to the messy reality of post-consumer plastic waste.
Polyolefins such as polyethylene and polypropylene make up a large share of global plastic production. They are widely used in packaging, consumer goods, household products and industrial applications. Chemical recycling routes that convert these long polymer chains back into shorter hydrocarbons could create feedstocks for new chemicals and fuels, complementing mechanical recycling and helping recover value from difficult waste streams.
The challenge is that mixed waste is hard to process cleanly. Even small amounts of PVC can be a major problem. Under hydrocracking conditions, PVC can release chlorine-containing species that poison or deactivate many standard catalysts, especially those based on noble metals and acidic zeolites. At the same time, the pore structure of traditional catalysts can be poorly matched to large polymer molecules and their bulky reaction intermediates. In narrow micropores, mass transport slows down and some active sites may be hard for polymer chains to reach.
The new work tackles both issues at once. The researchers show that tungsten carbide materials can provide closely spaced catalytic functions without relying on the classic noble metal plus zeolite design. In their system, W and W2C phases act as the “metal” sites, while hydroxyl groups associated with tungsten oxide species provide Brønsted acidity. Because these functions are built into the same material family, the active sites are naturally close together, which can improve reaction efficiency for large polymer substrates.
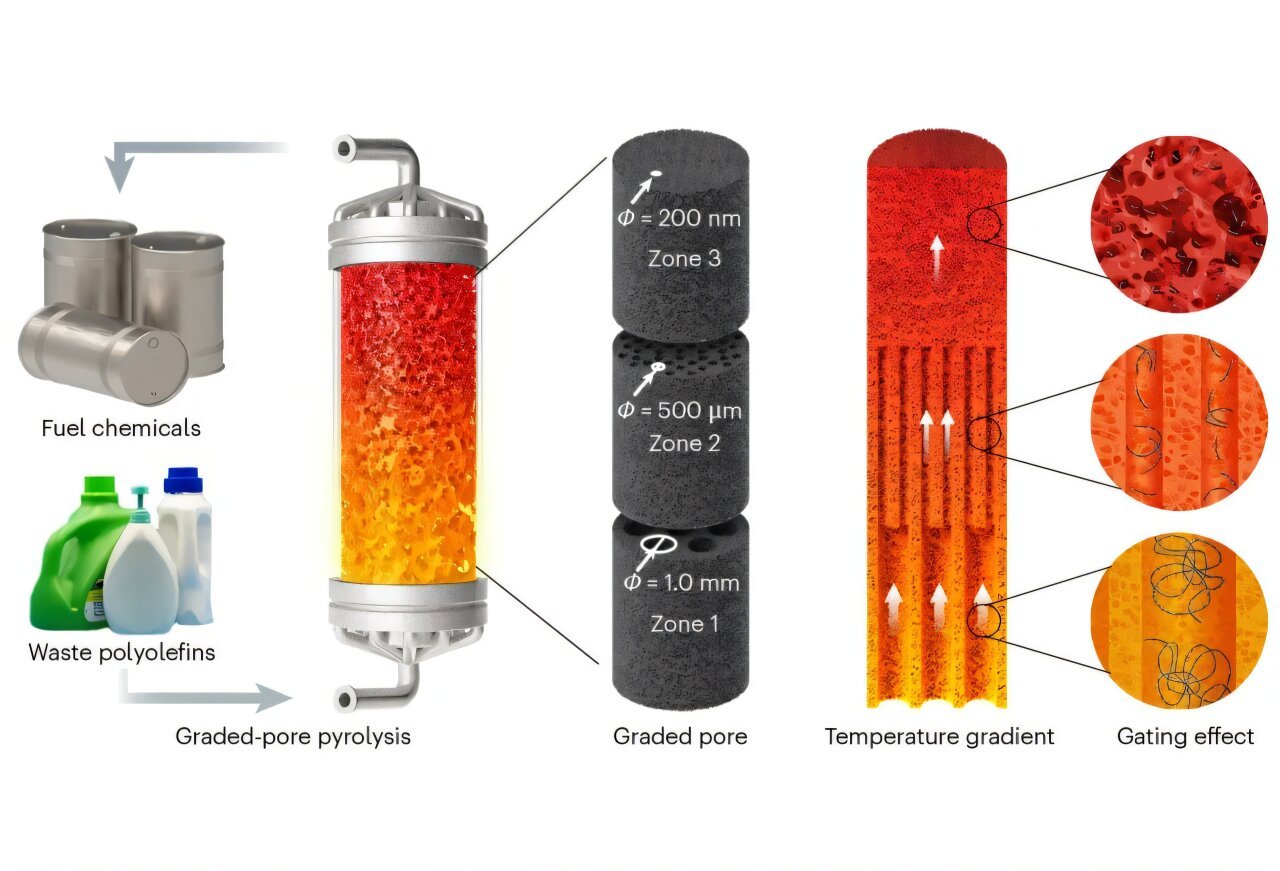
Why site proximity matters in polymer hydrocracking
For small hydrocarbons, bifunctional catalysis is already a well-known concept. A molecule may first undergo dehydrogenation on a metal site, then migrate to an acid site for skeletal rearrangement or bond scission and finally return to a metal site for hydrogenation. When the reactant is a long polymer chain rather than a small alkane, that choreography becomes more difficult. Large molecules move slowly, can become sterically hindered and may not easily access deep pore networks.
If the metal and acid functions are too far apart, the reactive intermediate formed at one site may not efficiently reach the other before unwanted side reactions occur. The new tungsten carbide system appears to reduce that problem by offering what the authors describe as unique site intimacy. In simple terms, the two required functions sit near each other on the catalyst, which is valuable for handling heavy polymer-derived species.
The team also found that the balance between metal-like and acid-like functions can be tuned by changing the carburization temperature during catalyst preparation. That temperature alters the relative amounts of tungsten carbide, metallic tungsten and tungsten oxide-related species. As a result, the ratio of metal sites to Brønsted acid sites can be adjusted.
This balance turned out to be critical. The study reports a volcano-shaped activity trend, meaning performance improved as the ratio approached an optimum and then declined when the balance shifted too far in either direction. That is a classic sign in catalysis that neither function alone is enough. Too few acid sites can limit cracking. Too few metal sites can slow the hydrogen transfer steps needed to keep the overall sequence moving efficiently and selectively.
A catalyst built for less-than-perfect plastic sorting
One of the most notable findings is the catalyst’s response to PVC contamination. Conventional hydrocracking catalysts can lose activity when chlorine is released from PVC under reaction conditions. That is a serious practical problem because waste sorting is expensive, imperfect and difficult at large scale. If a catalyst only works on highly purified polyolefin streams, its industrial usefulness may be limited.
In contrast, the tungsten carbide catalysts in this study maintained activity and in some cases even showed increased activity, when the polyolefin substrate contained 10 weight percent PVC. That result does not mean PVC suddenly becomes harmless in every recycling process and it does not remove the need for smart feed management. But it does suggest a much higher level of tolerance to chlorine-bearing contamination than is typical for established noble metal and zeolite systems.
That tolerance could be highly relevant for real municipal and commercial waste streams, where some level of cross-contamination is normal. A catalyst that can keep working in the presence of PVC could reduce preprocessing burdens, lower sorting costs, or widen the range of acceptable feedstocks for chemical recycling plants.
The use of tungsten is also significant from a materials standpoint. Noble metals such as platinum are effective catalytic components but can be expensive and vulnerable to poisoning. Transition-metal carbides have long attracted interest because they sometimes show catalytic behavior resembling that of precious metals while relying on more earth-abundant elements. This study extends that idea into polymer hydrocracking in a particularly compelling way.
Efficiency per acid site was markedly higher
The authors report that, on a per-Brønsted-acid-site basis, the tungsten carbide catalysts were more than an order of magnitude more efficient than conventional bifunctional catalysts. That is a striking comparison, because it suggests the gain is not simply due to having more active sites overall. Instead, each acid site appears to be used more effectively.
The researchers attribute much of that advantage to improved transport of polymer intermediates. In standard microporous catalysts, a meaningful fraction of potential activity may be inaccessible to large chains or hindered fragments. By contrast, the tungsten carbide system seems to present reactive functionality in a way that is easier for polymer-derived species to reach and use.
For the field of plastic chemical recycling, that detail matters. Polymer conversion is not just a question of intrinsic catalytic chemistry. Reactor performance often depends on whether large, viscous, or partially cracked molecules can physically access the sites needed for the next reaction step. A catalyst can look excellent in small-molecule testing yet struggle with real polymers if transport limitations dominate.
The kinetic data in the new study support a sequential cleavage mechanism in which each polyolefin chain is cut step by step rather than shattering randomly all at once. That behavior is consistent with broader understanding of hydrocracking chemistry. It also helps explain why the distribution of products and the concept of cracking ideality remain useful for thinking about polymers as well as smaller alkanes.
The authors note that trends in cracking ideality and selectivity followed those observed in short-alkane hydrocracking. That connection is valuable because it links polymer recycling chemistry to a more mature body of catalytic science. It suggests that design rules developed for refining and hydrocarbon processing may still apply, but they need to be adapted for the physical realities of polymer feedstocks.
What this means for the design of future recycling catalysts
The broader message of the work is that catalyst architecture matters as much as catalyst composition. Traditional bifunctional systems often combine separate materials to achieve metal and acid functionality. That approach can be powerful, but it can also create mismatches in site accessibility, diffusion path length and resistance to contaminants.
By building both functions into tungsten-containing carbide and oxide domains, the researchers created a catalyst family with several practical advantages. The metal-to-acid balance is tunable. The active functions are close together. The structure appears more compatible with large polymer-derived intermediates. And the system is notably more tolerant of chlorine released by PVC than conventional benchmarks.
This is especially important because the future of chemical recycling may depend less on ideal feedstocks and more on robust processing of imperfect ones. Industrial plants do not operate on textbook polymer samples. They process post-consumer films, mixed rigid packaging, contaminated industrial scrap and materials carrying labels, inks, fillers, additives and traces of other plastics. Catalysts that are highly active but fragile may not survive that environment.
A tougher catalyst platform could also improve process economics. If feed purification can be relaxed even slightly, the savings in sorting and pretreatment could be meaningful. If catalyst life improves in the presence of contaminants, shutdowns and replacement costs may fall. And if the catalyst converts polyolefins efficiently to shorter hydrocarbons under hydrogen, the output stream may be better suited for downstream upgrading into useful chemical products.
There are still questions to answer before such catalysts move toward industrial deployment. Long-term stability under continuous operation will matter. So will regeneration behavior, chlorine balance, product distributions under realistic mixed-plastic feeds and compatibility with additives and fillers commonly found in waste. Scale-up of catalyst synthesis and shaping into practical reactor forms will also be important.
Even so, the study gives the field a new direction. Rather than viewing PVC contamination only as a reason to avoid hydrocracking mixed plastics, it suggests that catalyst chemistry can be redesigned to cope with the problem. It also broadens the set of materials considered useful for bifunctional catalysis in polymer recycling, bringing transition-metal carbides into stronger focus.
That could inspire a wider search for catalyst families that combine metal and acid functions naturally, resist heteroatom poisoning and avoid the transport penalties associated with conventional microporous supports. Variants based on other carbide or oxycarbide systems may now draw more attention, especially if they can be tuned with the same level of control reported here.
For recycling researchers, the practical appeal is clear. Polyolefins remain the largest and most difficult-to-manage plastic fraction in many waste streams. Any technology that makes them easier to depolymerize or hydrocrack under realistic conditions could have major environmental and industrial value. A catalyst that stays active even when PVC is present addresses one of the most stubborn barriers to translating lab chemistry into real waste processing.
For catalysis scientists, the work is a reminder that multifunctional materials can outperform traditional combinations when site proximity and substrate size become dominant factors. The more complex the feed, the more important those design details become.
In that sense, tungsten carbide is not just another candidate catalyst. It may represent a broader strategy for building recycling systems around the actual composition of waste, rather than around idealized, perfectly sorted plastics. If that approach holds up under further testing, it could help make chemical recycling more tolerant, more flexible and more relevant to the plastics problem as it exists outside the laboratory.
The study appears under the title Intrinsically Bifunctional and Tunable Tungsten Carbide Catalysts Enable Efficient PVC-Compatible Polyolefin Hydrocracking. Its findings add a notable new option to the growing toolkit for polymer upcycling and feedstock recovery.
Related reading
More journal reading and science coverage connected to this topic.

DEVELOPMENT OF A RUBBER-BASED PRODUCT USING A MIXTURE EXPERIMENT: A CHALLENGING CASE STUDY
Title: DEVELOPMENT OF A RUBBER-BASED PRODUCT USING A MIXTURE EXPERIMENT: A CHALLENGING CASE STUDY Page Range: p.123-150 Author(s): Yahya Kaya; Piepel G F; Erdal…

EFFECT OF FLY ASH AS FILLER IN RUBBER – A COMPREHENSIVE STUDY OF THE VULCANISATE PROPERTIES OF STYRENE-BUTADIENE RUBBER COMPOUNDS
Title: EFFECT OF FLY ASH AS FILLER IN RUBBER - A COMPREHENSIVE STUDY OF THE VULCANISATE PROPERTIES OF STYRENE-BUTADIENE RUBBER COMPOUNDS Page Range: p.151-168…

CONTROLLABLE MICROWAVE-ASSISTED POLYCARBONATE WASTE CHEMICAL RECYCLING
Title: CONTROLLABLE MICROWAVE-ASSISTED POLYCARBONATE WASTE CHEMICAL RECYCLING Page Range: p.169-176 Author(s): Mir Mohammad Alavi Nikje; Askarzadeh M File size: Download the pdf (subscribers only)…
